Clinical Trials
Phase 3 trial
FESPIXON ® the 1st, large scale of RCT for a
M1/M2 macrophage regulating drug in DFUs patients

*Complete healing, defined as complete epithelialization maintained without drainage or requirement of dressings for at least 2 consecutive visits
DFUs = diabetic foot ulcers; RCT = randomized control trial; US = United States
Huang YY, et al. JAMA Netw Open. 2021 Sep 1;4(9):e2122607.
Baseline characteristics

*Due to previous diabetic foot ulcers
SD = standard deviation
Huang YY, et al. JAMA Netw Open. 2021 Sep 1;4(9):e2122607.
FESPIXON ® exhibited superior and faster efficacy in the healing of DFUs within only 16 weeks
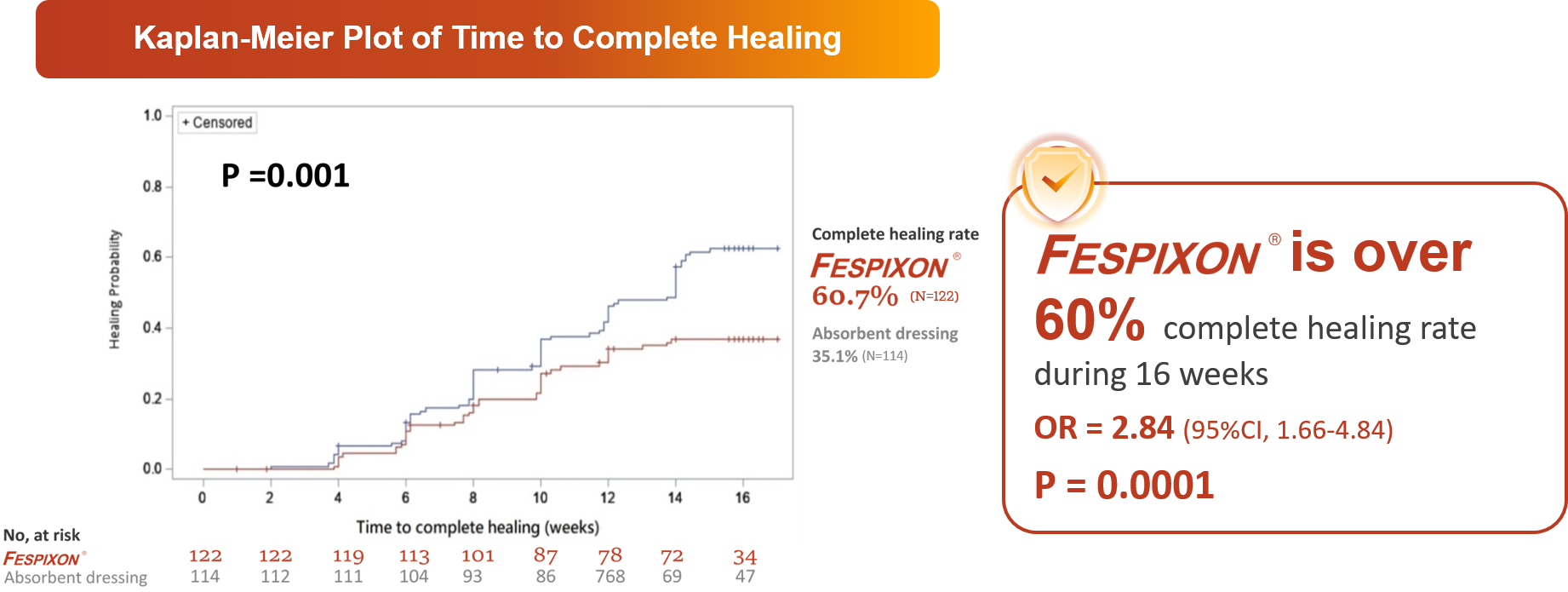
CI = confidence interval; DFUs = diabetic foot ulcers; OR = odds ratio; vs = versus
Huang YY, et al. JAMA Netw Open. 2021 Sep 1;4(9):e2122607.
FESPIXON ® demonstrated robust efficacy even in high-risk patients
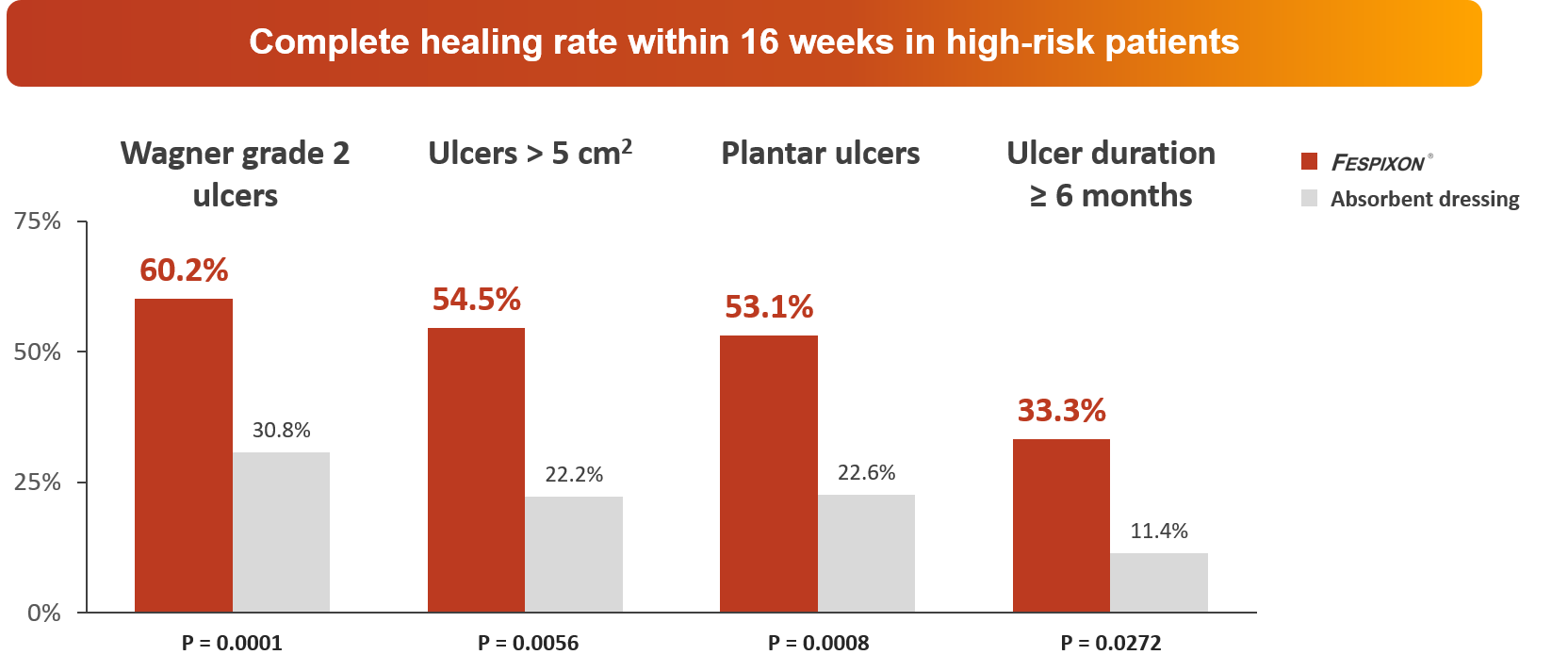
Huang YY, et al. JAMA Netw Open. 2021 Sep 1;4(9):e2122607.
FESPIXON ® offers robust safety profile

TEAEs = treatment-emergent adverse events
Huang YY, et al. JAMA Netw Open. 2021 Sep 1;4(9):e2122607.